Intracept® Procedure – Relief from Chronic Low Back Pain
A minimally invasive treatment for chronic low back pain
What Is Vertebrogenic Pain?
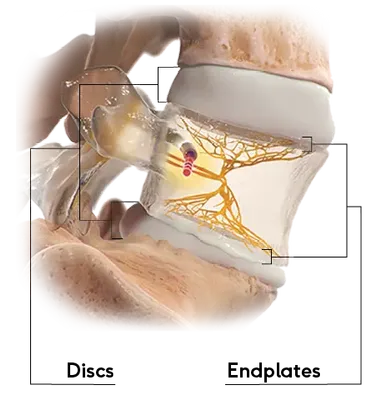
Vertebrogenic pain is a specific type of chronic low back pain that originates from damage to the vertebral endplates—the thin layers of tissue separating each spinal disc from the bones above and below. As these endplates deteriorate over time, often due to normal wear or disc degeneration, they can become inflamed and painful.
This pain is transmitted to the brain through the basivertebral nerve (BVN), which runs inside the vertebra. Unlike other types of back pain, vertebrogenic pain is well-defined and now treatable with targeted intervention.
Common Symptoms
You may be experiencing vertebrogenic pain if you have:
- ✓A deep, aching sensation in the center of your lower back
- ✓Discomfort that worsens with prolonged sitting or standing
- ✓Pain aggravated by bending, lifting, or physical activity
- ✓No significant radiating pain into the legs (unlike sciatica)
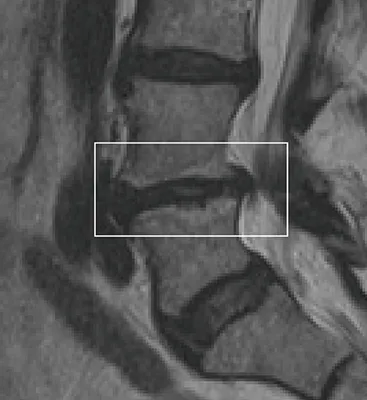
This condition is often accompanied by Modic changes—inflammatory changes visible on MRI that indicate endplate damage and are commonly associated with vertebrogenic pain.
What Is the Intracept® Procedure?
The Intracept® Procedure is a minimally invasive, outpatient treatment designed to stop pain at its source. By applying radiofrequency energy to the basivertebral nerve, the procedure disrupts pain signals—providing long-lasting relief without implants or major structural changes to the spine.
Key Benefits:
- ✓Implant-free and minimally invasive
- ✓Performed under anesthesia
- ✓Completed in under 60 minutes
- ✓No overnight hospital stay
- ✓Backed by rigorous clinical evidence for safety and long-term results
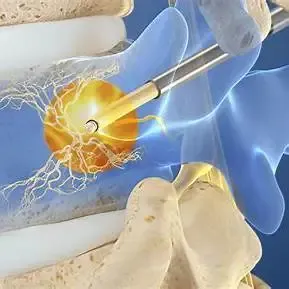
Quick Facts About the Intracept® Procedure
✅Same-day outpatient procedure
✅No spinal implants
✅Long-term relief after a single treatment
✅Proven safety with a low complication rate
✅Preserves your spine's natural structure
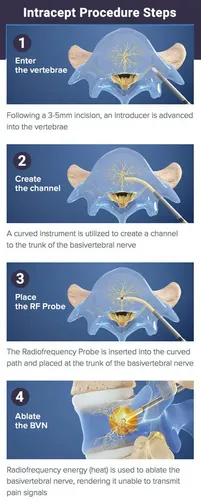
How the Procedure Works
1.A small probe is inserted into the affected vertebra using image guidance.
2.The basivertebral nerve is located within the bone.
3.Radiofrequency energy is delivered to ablate (disable) the nerve.
4.The small incision is closed, and patients typically return home the same day.
Long-Term Results You Can Trust
✅Nearly 80% of patients in a recent study said they would choose the procedure again¹
✅Less than 0.2% of serious device- or procedure-related complications reported¹
✅65% of patients returned to their pre-pain activity levels after 5 years²
✅Relief is supported by long-term, peer-reviewed clinical data
Are You a Candidate?
You may be eligible for the Intracept® Procedure if:
✓You've experienced chronic low back pain for six months or longer
✓You've tried physical therapy, medications, or injections without relief
✓Your MRI shows Modic changes, which indicate endplate inflammation
A personalized evaluation with our team will determine if this approach is right for your condition.
Contact Us – Serving Utah County
Let us help you find relief from chronic low back pain. Contact our team to schedule a consultation:
Fax
385-298-1009
Location
15 S. 1000 E. #250, Payson, UT 84651
The Intracept® Procedure is FDA-cleared and supported by long-term clinical data. Individual results may vary and should be discussed with your provider.
References
- Fischgrund, J. S., Rhyne, A., Macadaeg, K., Franke, J., Truumees, E., Kitchel, S., ... & Bae, H. (2020). Long-term outcomes following intraosseous basivertebral nerve ablation for the treatment of chronic low back pain: 5-year treatment arm results from a prospective randomized double-blind sham-controlled multi-center study. European Spine Journal, 29(8), 1925–1934.
- Koreckij, T., Kreiner, S., Khalil, J. G., Smuck, M., Markman, J., & Garfin, S. (2021). Prospective, randomized, multicenter study of intraosseous basivertebral nerve ablation for the treatment of chronic low back pain: 24-month treatment arm results. North American Spine Society Journal, 8, 100089.
- Relievant Medsystems. (2023). Clinical data on file. Retrieved from https://www.relievant.com/intracept/
- U.S. Food and Drug Administration. (2022). 510(k) Premarket Notification – Intracept® System. Retrieved from https://www.accessdata.fda.gov/
- Boston Scientific. (2023). Intracept® Procedure for vertebrogenic low back pain. Retrieved from https://www.bostonscientific.com/en-US/medical-specialties/pain-management/intracept.html